Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (2): 272-277.doi: 10.3969/j.issn.2095-4344.1514
Previous Articles Next Articles
Barrier materials for postoperative abdonimal adhesion: biological characteristics, merits and demerits
Yang Lili1, 2, Bian Yaoyao3, Zhao Min1, Wang Yetong3, Tang Shengjin3, Li Wenlin2, Zeng Li1, 2
- 1First Clinical Medicine College, Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China; 2Jingwen Library, Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China; 3School of Nursing, Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China
-
Received:2018-09-03Online:2019-01-18Published:2019-01-18 -
Contact:Zeng Li, Professor, Doctoral supervisor, First Clinical Medicine College, Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China; Jingwen Library, Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China -
About author:Yang Lili, Doctorate candidate, First Clinical Medicine College, Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China; Jingwen Library, Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81704084 (to YLL), 81373843 (to ZL), and 81673982 (to ZL); the Science
and Technology Projects of Jiangsu Provincial Bureau of Traditional Chinese Medicine, No. YB2017002 (to YLL), YB2015002 (to LWL); the
2018 Graduate Research and Practice Innovation Program of Jiangsu Province (General Program) No. 7 (to YLL); the Innovation and
Entrepreneurship Training Project of Nanjing University of Chinese Medicine in 2017, No. 16 (to TSJ and WYT)
CLC Number:
Cite this article
Yang Lili, Bian Yaoyao, Zhao Min, Wang Yetong, Tang Shengjin, Li Wenlin, Zeng Li. Barrier materials for postoperative abdonimal adhesion: biological characteristics, merits and demerits[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(2): 272-277.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
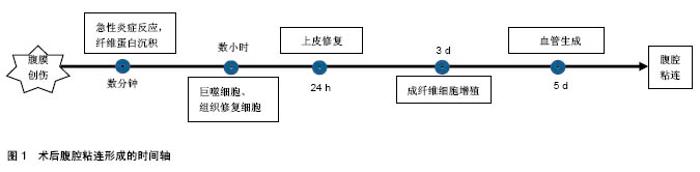
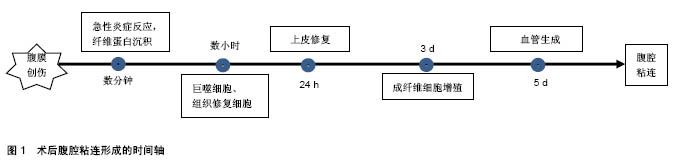
2.1 腹腔粘连的发病机制及相关因素 术后腹腔粘连通常是网膜、肠管、腹壁间的病理性粘连,这些连接可以是一层结缔组织的薄膜,或含有血管和神经组织的纤维“桥梁”,亦或是2个脏器表面的病理性连接。术后腹腔粘连起因于腹膜损伤,启动于炎症反应,爆发于局部修复,其病理生理过程迅速、级联且具有一定的复杂性。术后腹腔粘连的形成是一个与时俱进的过程,可分为4个阶段[9]:炎症阶段、纤溶阶段、纤维化阶段及重塑阶段。当腹膜损伤后,数分钟内局部出现急性炎症反应,毛细血管通透性增加、中性粒细胞和巨噬细胞迁移至受损部位形成纤维蛋白网;数小时内受损部位由巨噬细胞和“组织修复细胞”覆盖,而成纤维细胞及间皮细胞被血小板生长因子激活,迁移和增殖能力增强,纤溶活性增强,通常72 h内即可完成上皮修复;若72 h内不能完成修复过程,成纤维细胞第3天开始不断增殖并形成纤维蛋白网,同时局部血管第5天开始生成,二者结合最终形成致密粘连[10],见图1。"

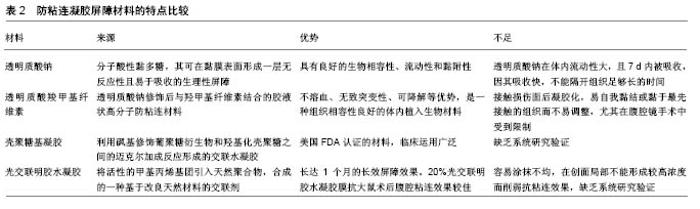
术后腹腔粘连形成过程相对复杂,从单一条索带导致循环障碍再到无症状性广泛致密粘连,多种危险因素影响粘连的形成,且粘连条索带的大小并不能预测粘连形成与否,因此正确认识粘连形成相关危险因素及粘连相关并发症,对正确辨别高风险患者至关重要,有助于采取有效的预防措施及对症治疗。 手术部位是首要因素之一,不同组织器官粘连的发生有显著差异[3,11-12],盆腔手术,如小肠、结肠、阑尾及子宫等部位的术后粘连发生率高于胃、胆囊、胰腺等部位。关于这种差异,有学者归因于整合素,腹膜脏器与浆液组织之间存在不同数量的整合素,其是细胞外基质Ca2+依赖性黏附分子,是促黏附重要因素,也是不同组织器官粘连发生潜在机制之一。组织损伤引起炎症反应,并且在整合素较高的组织中聚集,如子宫和小肠浆膜中整合素ανβ3显著高于其他腹腔脏器中整合素水 平[13]。手术类型也是粘连发生的重要相关因素之一,腹腔镜手术能够减少45%-50%粘连的发生[5,14-15],但仍然不能避免其发生。缝线也是诱发粘连形成重要因素之一,据统计25%患者手术创面出现缝线肉芽肿[16],粘连的形成与缝线规格有直接联系,单丝缝合线对腹膜损伤较小,故能降低粘连的发生率,而粗直径缝合线产生较大炎症刺激,会加重感染风险。同时年龄也可能是术后腹腔粘连的相关因素,既往5年内有腹腔或盆腔手术史且年龄低于60岁,因粘连相关疾病再入院风险会增加1 倍[3]。此外,急诊手术也被认为是粘连形成过程中的危险因素,Sisodia等[17]报道急诊手术发生粘连的相关并发症显著高于选择性外科手术。 2.2 腹腔粘连的防治措施 术后腹腔粘连有效防治一直是亟待解决的临床问题,针对复杂的术后粘连病理机制及众多危险因素,手术技巧及屏障材料日趋成为防治术后预防粘连研究关注点。 2.2.1 手术技巧 腹腔粘连与术中腹膜损伤密切相关,腹膜损伤起因于手术创伤,因此避免腹膜创伤是减少术后腹腔粘连形成的可行措施之一。术中精细操作,精准止血、适当冲洗、充分引流,是减少组织创伤、炎症及防治浆膜损伤至关重要的手段[18]。此外,术中减少肠管暴露,防止肠道表面干燥,避免外源性异物残留,可减少粘连发生[18-19]。相较于使用聚丙烯缝合线或钛合金固定器等非吸收性固定方法,可吸收缝线术后粘连形成率更低[20-21]。研究发现剖宫术后缝合腹直肌会导致较少的纤维条索和致密粘连,因此术中可适当考虑缝合腹直肌,以减少粘连的发生[22]。 与传统开腹手术相比,腹腔镜手术可减少损伤,减少术后腹腔粘连的发生,已被一些临床研究所证实[23-24]。需要关注的是腹腔镜手术可减少但不能消除腹壁与脏器之间的粘连,且不能减少内脏粘连的发生率[25]。另有报道显示,腹腔镜手术在预防粘连方面与开腹手术相比无显著性优势[26-27]。因此腹腔镜手术在术后粘连的关系亟待深入了解,腹腔镜手术治疗粘连的疗效评估亟待肯定。 2.2.2 术后腹腔粘连防治屏障材料 近年来术后腹腔粘连防治屏障材料逐步得到认可并被广泛应用,防粘连材料能够通过物理屏障方式在粘连形成的早期阶段使得受创的区域和外周组织隔离,防止腹腔粘连形成。防粘连材料目前从制剂类型分析,主要存在溶液剂、凝胶剂及膜剂3种剂型,见表1-3。通过对腹腔粘连防治屏障材料进行分析比较,探讨各类防粘连材料的优势与不足,以期为临床防治和科学研究提供参考,为新型防粘连屏障材料的研发与转化提供思路。 溶液屏障材料:透明质酸钠-磷酸缓冲盐是透明质酸钠与磷酸缓冲盐结合的高分子溶液。动物实验发现,术中使用透明质酸钠-磷酸缓冲盐可有效减少浆膜损伤、炎症,降低术后粘连。临床实验发现在复杂的妇科盆腔手术中,透明质酸钠-磷酸缓冲盐可显著降低不同部位新生粘连的发生率,并降低其严重程度,无毒副作用[28]。但因其为液态,具有向低处流动特点,在临床实际应用过程中受患者体位改变而影响治疗效果。 N,O-羧甲基壳聚糖是基于壳聚糖的水溶性衍生物,是最常见的壳聚糖基溶液防粘连材料之一。Wang等[29]发现N,O-羧甲基壳聚糖可以阻断炎性细胞在损伤部位聚集并减轻局部炎症反应,降低手术部位的纤维化反应,减少粘连形成。Zhou等[30]研究同样发现N,O-羧甲基壳聚糖可抑制损伤部位成纤维细胞和巨噬细胞黏附、沉积及随后的粘连形成。对于N,O-羧甲基壳聚糖的防粘连机制,目前的研究还未完全阐明,且N,O-羧甲基壳聚糖在腹腔容易被体液稀释,容易流动到其他部位或不易在局部形成较高浓度而失效。 4%葡聚糖溶液是美国食品和药物管理局接受并认证的防粘连产品,其在临床研究中广泛使用。Brown等[31]对4%葡聚糖溶液减少腹腔镜术后粘连的效果和安全性评价,发现4%葡聚糖溶液在粘连形成关键时期对损伤修复过程进行阻断,即术后2-7 d通过腹腔进行扩散,可在腹腔内存留4 d,能有效分隔受损面,防治术后腹腔粘连的发生。但4%葡聚糖溶液在手术部位会引起液体渗漏,患者偶有不适感及尿潴留等不良反应[32-33]。 凝胶屏障材料:透明质酸钠是一种大分子酸性黏多糖,其可在黏膜表面形成一层无反应性且易于吸收的生理性屏障,具有良好的生物相容性、流动性和黏附性。在浆膜表面涂抹透明质酸钠,可一定程度减少腹膜擦伤或干燥引起的间接损伤,但对损伤后组织无效[28]。透明质酸钠可降低轻度粘连发生率,减少粘连评分低的粘连,对粘连范围小的术区粘连有效,而对中、重度粘连并不适用[34]。一般来说,透明质酸钠在体内流动性大,且7 d内被吸收,因其吸收快,不能隔开组织足够长的时间,难以在粘连形成的关键阶段预防粘连发生,故而作为腹膜修复期形成的一种暂时性屏障。自交联透明质酸钠凝胶是利用发酵技术将普通透明质酸钠分子进行独特的交联、改性后形成的透明质酸团块,保留了透明质酸钠的基本生化特性,在体内降解时间延长,因其体内滞留时间长,故炎症反应较轻[35]。研究证实,自交联透明质酸钠凝胶具有良好的动力黏稠度及组织相容性,能较好地预防术后粘连的发生,安全性好[36-37]。 透明质酸羧甲基纤维素是透明质酸钠修饰后与羟甲基纤维素结合的胶液状高分子防粘连材料,其可降低成纤维细胞对纤连蛋白的黏附,并可阻隔浆膜面与覆膜面接触长达7 d,具有不溶血、无致突变性、可降解等优势,是一种组织相容性良好的体内植入生物材料。多项前瞻性研究及荟萃分析均表明,透明质酸羧甲基纤维素可安全有效防止术后粘连的形成,减少发生范围和严重程 度[38-40]。但是透明质酸羧甲基纤维素接触损伤面后凝胶化,易自我黏结或黏于最先接触的组织而不易调整,尤其在腹腔镜手术中受到限制。改良后的透明质酸羧甲基纤维素可以弥补这一缺点,将其以粉末形式喷洒在微创手术远端创面,动物实验及临床试验均证实改良后的透明质酸羧甲基纤维素能有效减少粘连的发生[41-42]。 壳聚糖基凝胶(壳聚糖/葡聚糖共混原位交联凝胶)是利用砜基修饰葡聚糖衍生物和羟基化壳聚糖之间的迈克尔加成反应形成的交联水凝胶。目前已经被研究的交联凝胶主要有氧化葡聚糖/N-羧乙基壳聚糖、氧化葡聚糖水凝胶/羧甲基壳聚糖及氧化葡聚糖水凝胶/硫醇化壳聚糖等[43]。研究发现,在腹腔镜及开放性手术后动物模型中应用壳聚糖/葡聚糖凝胶,可显著减少盲肠磨损及肠切口处粘连且对伤口愈合无不利影响[44]。虽然壳聚糖和葡聚糖均是通过美国FDA认证的材料,被证实具有良好的生物相容性,且研究证实该原位交联凝胶防粘连效果与防粘连膜Seprafilm®相当,但还需要大量的动物腹腔粘连模型来评估防粘连效果。 光交联明胶水凝胶是将活性的甲基丙烯基团引入天然聚合物,合成的一种基于改良天然材料的交联剂,具有长达1个月的长效屏障效果。研究发现,20%光交联明胶水凝胶膜抗大鼠术后腹腔粘连的效果较佳[45]。虽然该材料研究在动物模型上取得一定成果,但还处于实验阶段,后续还要大量实验进行验证研究。另外,在使用该水凝胶过程中可能会出现涂抹不均,在创面局部不能形成较高浓度,削弱了抗粘连效果。 膜屏障材料:基于光反应性的壳聚糖衍生物,O-羧甲基壳聚糖防粘连膜具有高水溶性、非细胞毒性及光引发固化反应,并具有良好生物相容性和细胞抗黏附作用。固化的O-羧甲基壳聚糖防粘连膜可在组织内停留 5 d,其抗粘连性能堪比透明质酸羧甲基纤维素。随着对载药壳聚糖防粘连膜的不断研究,壳聚糖防粘连膜抗粘连潜能正被不断挖掘,但距离临床转化还需进一步研究,主要存在膜的机械强度不够大、生物黏附强度不够,降解可控性不足等缺点[46]。 聚乳酸防粘连膜是聚乳酸树脂高分子纳米薄膜,是美国FDA首批认证并批准的防粘连材料,具有促进组织修复、无异物反应、无毒副作用、能有效隔离创面、保证隔离时间等优点。产妇术后应用聚乳酸防粘连膜,可预防盆腹腔粘连,对产妇的二次剖宫术有积极作用[47]。动物实验发现,聚乳酸防粘连膜抗粘连效果优于透明质酸钠[48]。但该材料为透明薄膜,需根据创面大小裁剪,常需缝线固定,对较深病灶使用不方便且难以在微创手术中使用,降解缓慢,术后2周仍保持完整性。 氧化再生纤维素是经NO处理的可吸收人造纤维补片,其可在腹腔内使用8 h后转化为黏稠的凝胶状物质,覆盖在受损腹膜表面并在2周内彻底吸收。荟萃分析表明,开腹及腹腔镜手术中使用氧化再生纤维素,能显著减少首次粘连和再次粘连发生率[49]。但氧化再生纤维素的缺点是遇血时效能下降,因此使用时需仔细操作止血且不适用于腹腔镜手术。美国FDA MAUDE数据库显示其有52例不良反应报道,主要表现为感染、不完全吸收、压迫神经并造成疼痛不适等。虽然氧化再生纤维素在临床应用已有十多年,但其生物安全性还有待进一步评价。 静电纺丝膜是利用静电纺丝技术制备的纳米纤维抗粘连材料,为近年来世界材料科学技术领域的研究热点之一。Wang等[50]的研究发现,利用静电纺丝技术制备预防腹部手术的可吸收超细纤维防粘连膜,具有从纳米纤维支架释放活性药物而不失结构完整性和功能性的特点。Li等[51]采用静电纺丝技术制备聚乳酸-乙醇酸共聚物/聚乙二醇可吸收电纺聚合膜,发现其可有效预防大鼠术后腹腔粘连,具有良好的生物降解性。Chang等[52]利用静电纺丝技术制备出具有芯鞘结构的壳聚糖/海藻酸钠纤维毡,并分别使用体外细胞黏附模型和动物模型评估该纤维毡的组织抗粘连作用,发现其可有效减少组织粘连的形成。 "

| [1] Brochhausen C, Schmitt VH, Mamilos A, et al. Expression of CD68 positive macrophages in the use of different barrier materials to prevent peritoneal adhesions-an animal study. J Mater Sci Mater Med. 2017;28(1):15. [2] Ray NF, Denton WG, Thamer M, et al. Abdominal adhesiolysis: inpatient care and expenditures in the United States in 1994. J Am Coll Surg. 1998;186(1):1-9. [3] Parker MC, Wilson MS, Menzies D, et al. The SCAR-3 study: 5-year adhesion-related readmission risk following lower abdominal surgical procedures. Colorectal Dis. 2005;7(6):551-558. [4] Tingstedt B, Johansson J, Nehez L, et al. Late abdominal complaints after appendectomy--readmissions during long-term follow-up. Dig Surg. 2004;21(1):23-27. [5] Schnuriger B, Barmparas G, Branco BC, et al. Prevention of postoperative peritoneal adhesions: a review of the literature. Am J Surg. 2011;201(1):111-121. [6] Brochhausen C, Schmitt VH, Planck CN, et al. Current strategies and future perspectives for intraperitoneal adhesion prevention. J Gastrointest Surg. 2012;16(6):1256-1274. [7] Ward BC, Panitch A. Abdominal adhesions: current and novel therapies. J Surg Res. 2011;165(1):91-111. [8] Ten BR, Stommel M, Strik C, et al. Benefits and harms of adhesion barriers for abdominal surgery: a systematic review and meta-analysis. Lancet. 2014;383(9911):48-59. [9] 白景瑞,吴咸中.术后腹腔粘连的发生机制及其防治[J].中国中西医结合外科杂志,2011,17(3):332-334.[10] Koninckx PR, Gomel V, Ussia A, et al. Role of the peritoneal cavity in the prevention of postoperative adhesions, pain, and fatigue. Fertil Steril. 2016;106(5):998-1010. [11] Ten BR, Issa Y, van Santbrink EJ, et al. Burden of adhesions in abdominal and pelvic surgery: systematic review and met-analysis. BMJ. 2013;347:f5588. [12] Lower AM, Harn RJ, Clark D, et al. Adhesion-related readmissions following gynaecological laparoscopy or laparotomy in Scotland:an epidemiological study of 24 046 patients. Hum Reprod. 2004;19(8): 1877-1885. [13] Fletcher NM, Awonuga AO, Abusamaan MS, et al. Adhesion phenotype manifests an altered metabolic profile favoring glycolysis. Fertil Steril. 2016;105(6):1628-1637. [14] Ouaissi M, Gaujoux S, Veyrie N, et al. Post-operative adhesions after digestive surgery: their incidence and prevention: review of the literature. J Visc Surg. 2012;149(2):e104-e114. [15] Lauder CI, Garcea G, Strickland A, et al. Abdominal adhesion prevention: still a sticky subject? Dig Surg. 2010;27(5):347-358. [16] Kosan M, Gonulalan U, Ozturk B, et al. Tissue reactions of suture materials (polyglactine 910, chromed catgut and polydioxanone) on rat bladder wall and their role in bladder stone formation. Urol Res. 2008;36(1):43-49. [17] Sisodia V, Sahu SK, Kumar S. Clinical Profile of Patients with Postoperative Adhesive Intestinal Obstruction and its Association with Intraoperative Peritoneal Adhesion Index. Chirurgia(Bucur). 2016; 111(3):251-258. [18] Ten BR, Kok-Krant N, Bakkum EA, et al. Different surgical techniques to reduce post-operative adhesion formation: a systematic review and meta-analysis. Hum Reprod Update. 2013;19(1):12-25. [19] Corona R, Verguts J, Koninckx R, et al. Intraperitoneal temperature and desiccation during endoscopic surgery. Intraoperative humidification and cooling of the peritoneal cavity can reduce adhesions. Am J Obstet Gynecol. 2011;205(4):391-392. [20] Schreinemacher M H, van Barneveld K W, Peeters E, et al. Adhesions to sutures, tackers, and glue for intraperitoneal mesh fixation: an experimental study. Hernia. 2014;18(6):865-872. [21] Ishikawa K, Sadahiro S, Tanaka Y, et al. Optimal sutures for use in the abdomen: an evaluation based on the formation of adhesions and abscesses. Surg Today. 2013;43(4):412-417. [22] Lyell DJ, Caughey AB, Hu E, et al. Rectus muscle and visceral peritoneum closure at cesarean delivery and intraabdominal adhesions. Am J Obstet Gynecol. 2012;206(6):511-515. [23] Burns EM, Currie A, Bottle A, et al. Minimal-access colorectal surgery is associated with fewer adhesion-related admissions than open surgery. Br J Surg. 2013;100(1):152-159. [24] Smolarek S, Shalaby M, Paolo AG, et al. Small-Bowel Obstruction Secondary to Adhesions After Open or Laparoscopic Colorectal Surgery. JSLS. 2016;20(4). pii: e2016.00073.doi:10.4293/JSLS. 2016. 00073. [25] Sidana A, Hernandez DJ, Feng Z, et al. Treatment decision-making for localized prostate cancer: what younger men choose and why. Prostate. 2012;72(1):58-64. [26] Scholin J, Buunen M, Hop W, et al. Bowel obstruction after laparoscopic and open colon resection for cancer: results of 5 years of follow-up in a randomized trial. Surg Endosc. 2011;25(12):3755-3760. [27] Mais V. Peritoneal adhesions after laparoscopic gastrointestinal surgery. World J Gastroenterol. 2014;20(17):4917-4925. [28] Liakakos T, Thomakos N, Fine PM, et al. Peritoneal adhesions: etiology, pathophysiology, and clinical significance. Recent advances in prevention and management. Dig Surg. 2001;18(4):260-273. [29] Dong W, Han B, Feng Y, et al. Pharmacokinetics and biodegradation mechanisms of a versatile carboxymethyl derivative of chitosan in rats: in vivo and in vitro evaluation. Biomacromolecules. 2010, 11(6):1527-1533. [30] Zhou J, Lee JM, Jiang P, et al. Reduction in postsurgical adhesion formation after cardiac surgery by application of N, O-carboxymethyl chitosan. J Thorac Cardiovasc Surg. 2010;140(4):801-806. [31] Brown CB, Luciano AA, Martin D, et al. Adept (icodextrin 4% solution) reduces adhesions after laparoscopic surgery for adhesiolysis: a double-blind, randomized, controlled study. Fertil Steril. 2007;88(5): 1413-1426. [32] Yeo Y, Kohane DS. Polymers in the prevention of peritoneal adhesions. Eur J Pharm Biopharm. 2008;68(1):57-66[33] Nesbitt-Hawes EM, Zhang CS, Won HR, et al. Urinary retention following laparoscopic gynaecological surgery with or without 4% icodextrin anti-adhesion solution. Aust N Z J Obstet Gynaecol 2013;53(3):305-309. [34] Liu H, Xu Y, Yi N, et al. Efficacy and Safety of Hyaluronic Acid Gel for the Prevention of Intrauterine Adhesion: A Meta-Analysis of Randomized Clinical Trials. Gynecol Obstet Invest. 2018;83(3):227-233. [35] Chen Q, Sun G, Wang Y, et al. The evaluation of two new hyaluronan hydrogels as nasal dressing in the rabbit maxillary sinus. Am J Rhinol Allergy. 2012;26(2):152-156. [36] 梁文锴,郭全义,韩树峰,等.Micro-CT联合硬膜外造影在医用自交联透明质酸钠凝胶预防椎板切除后硬膜外粘连中的应用[J].中国组织工程研究, 2012,16(43):8094-8099.[37] 夏棣萍,陈开拉,陈华.自交联透明质酸钠凝胶在中重度宫腔粘连行宫腔镜手术后患者中的应用效果[J].中国内镜杂志, 2016,22(3):68-71.[38] Beck DE, Cohen Z, Fleshman JW, et al. A prospective, randomized, multicenter, controlled study of the safety of Seprafilm adhesion barrier in abdominopelvic surgery of the intestine. Dis Colon Rectum. 2003; 46(10):1310-1319. [39] Kusunoki M, Ikeuchi H, Yanagi H, et al. Bioresorbable hyaluronate- carboxymethylcellulose membrane (Seprafilm) in surgery for rectal carcinoma: a prospective randomized clinical trial. Surg Today. 2005; 35(11):940-945. [40] Zeng Q, Yu Z, You J, et al. Efficacy and safety of Seprafilm for preventing postoperative abdominal adhesion: systematic review and meta-analysis. World J Surg. 2007;31(11):2125-2131, 2132. [41] Sheldon HK, Gainsbury ML, Cassidy MR, et al. A sprayable hyaluronate/carboxymethylcellulose adhesion barrier exhibits regional adhesion reduction efficacy and does not impair intestinal healing. J Gastrointest Surg. 2012;16(2):325-333. [42] Fossum GT, Silverberg KM, Miller CE, et al. Gynecologic use of Sepraspray Adhesion Barrier for reduction of adhesion development after laparoscopic myomectomy: a pilot study. Fertil Steril. 2011;96(2):487-491. [43] Falabella CA, Melendez MM, Weng L, et al. Novel macromolecular crosslinking hydrogel to reduce intra-abdominal adhesions. J Surg Res. 2010;159(2):772-778. [44] Lauder C, Strickland A, Maddern G. Use of a modified chitosan-dextran gel to prevent peritoneal adhesions in a porcine hemicolectomy model. J Surg Res. 2011;171(2):877-882. [45] Wu W, Ni Q, Xiang Y, et al. Fabrication of a photo-crosslinked gelatin hydrogel for preventing abdominal adhesion. Rsc Adv. 2016;6(95): 92449-92453. [46] 李纪伟,贺金梅,尉枫,等.壳聚糖基防粘连材料的构建及应用进展[J].高分子通报,2016,29(2):86-93.[47] 王东营,李冬,邹颖刚,等.防粘连措施在妇产科手术中的应用[J].中国妇幼保健,2018,33(2):461-464.[48] Lin LX, Yuan F, Zhang HH, et al. Evaluation of surgical anti-adhesion products to reduce postsurgical intra-abdominal adhesion formation in a rat model. PLoS One. 2017;12(2):e172088. [49] Ahmad G, O'Flynn H, Hindocha A, et al. Barrier agents for adhesion prevention after gynaecological surgery. Cochrane Database Syst Rev. 2015;(4):D475. [50] Wang H, Li M, Hu J, et al. Multiple targeted drugs carrying biodegradable membrane barrier: anti-adhesion, hemostasis, and anti-infection. Biomacromolecules. 2013;14(4):954-961. [51] Li J, Zhu J, He T, et al. Prevention of intra-abdominal adhesion using electrospun PEG/PLGA nanofibrous membranes. Mater Sci Eng C Mater Biol Appl. 2017;78:988-997. [52] Chang JJ, Lee YH, Wu MH, et al. Electrospun anti-adhesion barrier made of chitosan alginate for reducing peritoneal adhesions. Carbohydr Polym. 2012;88(4):1304-1312. [53] Parker MC. Epidemiology of adhesions: the burden. Hosp Med. 2004; 65(6):330-336. [54] Arung W, Meurisse M, Detry O. Pathophysiology and prevention of postoperative peritoneal adhesions. World J Gastroenterol. 2011; 17(41):4545-4553. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Li Jun, Zuo Xinhui, Liu Xiaoyuan, Zhang Kai, Han Xiangzhen, He Huiyu, . Effect of over expression of miR-378a on osteogenic and vascular differentiation of bone marrow mesenchymal stem cell sheet [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4939-4944. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||